FastGro Fertiliser Plant
The Sunshine Coast Regional Council has asked for a study on the effect of a newly proposed 'FASTGRO' fertiliser company on water quality and surrounding wildlife. 'FASTGRO' will produce a range of byproducts and pollutants including:
Two hypothetical test were conducted on a water sample to conduct the effect of certain variables on the quality of water. Two test included the release of hot water and the release of Sodium Chloride (Salt).
Temperature can have a detrimental effect on water quality by dropping dissolved oxygen levels because the water is less capable to hold oxygen. Low dissolved oxygen levels leave aquatic plants and animals more susceptible to disease, parasites and other pollutants.
Sodium Chloride (NaCl), most commonly know as salt, is an ingredient in some fertilisers and the chances of accidental spills or dumping is highly likely. Salt is highly soluble and this makes it extremely easy for it to effect water quality. High concentrations of salt can cause many plants to become unhealthy and die and can also cause erosion. Salt levels also effect the conductivity of water. When dissolved, Sodium Chloride (NaCl) become Sodium ions (Na+) and Chlorine ions (Cl-), each conducting electricity. Therefore, the more Sodium Chloride present in water, the higher the conductivity.
- Nitrates
- Phosphates
- Nutrients causing Eutrophication
- Sodium Chloride (salt)
- Hot water
- Ammonia
- Acids
Two hypothetical test were conducted on a water sample to conduct the effect of certain variables on the quality of water. Two test included the release of hot water and the release of Sodium Chloride (Salt).
Temperature can have a detrimental effect on water quality by dropping dissolved oxygen levels because the water is less capable to hold oxygen. Low dissolved oxygen levels leave aquatic plants and animals more susceptible to disease, parasites and other pollutants.
Sodium Chloride (NaCl), most commonly know as salt, is an ingredient in some fertilisers and the chances of accidental spills or dumping is highly likely. Salt is highly soluble and this makes it extremely easy for it to effect water quality. High concentrations of salt can cause many plants to become unhealthy and die and can also cause erosion. Salt levels also effect the conductivity of water. When dissolved, Sodium Chloride (NaCl) become Sodium ions (Na+) and Chlorine ions (Cl-), each conducting electricity. Therefore, the more Sodium Chloride present in water, the higher the conductivity.
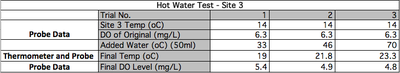
test 1 - Release of Hot Water
Hypothesis: The release of hot water into Coochin Creek will cause Dissolved Oxygen (DO) levels to decrease. Also, the higher temperatures will cause the Biological Oxygen Demand (BOD) to increase which will in turn decrease DO further.
Results:
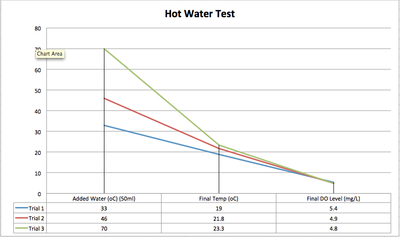
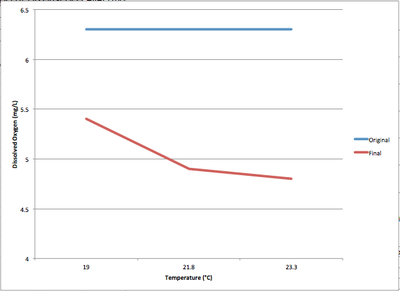
It was hypothesised that the release of hot water would cause DO levels to decrease. As seen in Graph 1b, hot water spills would cause a considerable drop in Dissolved Oxygen levels. In Table 1a, water temperatures added reached up to 70°C and the sample water temperatures only rose from 14°C to 23.3°C. These levels of dissolved oxygen are low however the water is not considered polluted until it drops to under 4mg/L.
Results:
It was hypothesised that the release of hot water would cause DO levels to decrease. As seen in Graph 1b, hot water spills would cause a considerable drop in Dissolved Oxygen levels. In Table 1a, water temperatures added reached up to 70°C and the sample water temperatures only rose from 14°C to 23.3°C. These levels of dissolved oxygen are low however the water is not considered polluted until it drops to under 4mg/L.
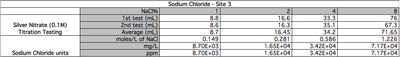
Test 2 - Release of sodium Chloride (salt)
Hypothesis: As the percentage of Sodium Chloride (NaCl) gets higher the value of ppm of NaCl will also rise in a linear fashion.
Results:
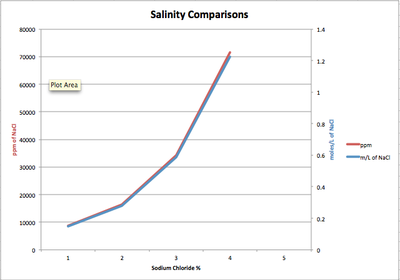
The salinity of water was tested by adding small percentages of sodium chloride to water samples. In Table 3, Site 3 has a value of 8.56x10^2 moles per litre of Sodium Chloride. In Graph 2a, the percentage of salinity doubles each time and the ppm increases exponentially. Water thats is <2000ppm is usually used for water irrigation and is still considered fresh. Salt water is usually 30,000-50,000ppm. The test shows that a percentage of salt of just 3% would be harmful to the fresh water environment.
Results:
The salinity of water was tested by adding small percentages of sodium chloride to water samples. In Table 3, Site 3 has a value of 8.56x10^2 moles per litre of Sodium Chloride. In Graph 2a, the percentage of salinity doubles each time and the ppm increases exponentially. Water thats is <2000ppm is usually used for water irrigation and is still considered fresh. Salt water is usually 30,000-50,000ppm. The test shows that a percentage of salt of just 3% would be harmful to the fresh water environment.
Conclusion
The affects of hot water and sodium chloride from a fertiliser plant would have diverse effects on water quality and the environment.
If hot water were to leak or be dumped from a hot water plant, the amount and temperature would only be small and would not make much of a difference to the water quality of Coochin Creek. However if these amounts were to be large and the temperature was over 70°C, then this might have a negative effect on the water and its inhabitants.
Sodium Chloride (NaCl) is only used in fertilisers in very small amounts. For 'FASTGRO' to pollute Coochin Creek with NaCl, it would have to be released directly or in very strong amounts in any byproducts. Tests showed that only a very small percentage of NaCl would be enough to change the fresh water of the site. However, because sodium chloride is not used in fertilisers in very large amounts, any contaminants would be in small quantities and not effect the water or the environment greatly.
If hot water were to leak or be dumped from a hot water plant, the amount and temperature would only be small and would not make much of a difference to the water quality of Coochin Creek. However if these amounts were to be large and the temperature was over 70°C, then this might have a negative effect on the water and its inhabitants.
Sodium Chloride (NaCl) is only used in fertilisers in very small amounts. For 'FASTGRO' to pollute Coochin Creek with NaCl, it would have to be released directly or in very strong amounts in any byproducts. Tests showed that only a very small percentage of NaCl would be enough to change the fresh water of the site. However, because sodium chloride is not used in fertilisers in very large amounts, any contaminants would be in small quantities and not effect the water or the environment greatly.